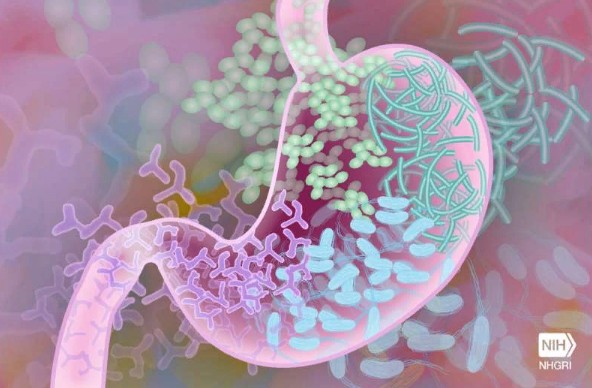
A new study published today in Nature Genetics sheds light on the impact of our genes on our gut health and their involvement in determining the composition of our gut flora.
We’ve long known that what we consume has a significant impact on the composition of our gut microbiota, but the role of our own genes is still unknown.
This new study finds a substantial link between the prevalence of specific microorganisms in our gut and the presence of certain genes that vary by population, including genes that are closely linked to disorders like lactose intolerance and blood type.
The authors used high-performance computing to analyze large genomic and microbiome datasets, led by the Cambridge-Baker Systems Genomics Initiative, which included researchers from the Baker Heart and Diabetes Institute in Melbourne and the University of Cambridge in the United Kingdom, as well as researchers from the Universities of Helsinki, Turku (Finland), and the University of California San Diego (USA).
Previous research on how human genetic variation affects gut microbiota has shown an ambiguous and complex interaction between environment and genetics, which the authors attempted to elucidate with a large sample size dataset.
Researchers analyzed the microorganisms in the gut, dietary information, and health records of nearly 6000 people from various geographical areas of Finland over a 16-year period using a large-scale population cohort dataset from the internationally renowned FINRISK Study (a large Finnish population survey).
Dr. Guillaume Meric, a Baker Institute researcher and microbiologist who co-led the study, said it was one of the largest of its kind and provided statistically robust findings from a large representative population survey, allowing for a better understanding of the role of genes on our gut microbiota.
Huge computer capabilities paired with more affordable sequencing for a large number of samples, according to Dr. Meric, have made this a hot area of study, with several comparable high-impact studies being published, or about to be published.
He explained that the researchers were able to characterize human genetic variations associated with varying concentrations and diversity of microbes in the gut, as well as predict possible causative or protective links between specific gut microbes and various diseases, thanks to the deep data dive.
According to Dr. Meric, the study found a number of extremely significant links between genes, gut microbes, and a variety of diseases and disorders, with intriguing candidates associated with lactose intolerance, colorectal cancer, and major depressive disorder.
The researchers discovered 567 different genetic variations linked to levels of more than 200 bacteria species in the gut, and the publication opens up new avenues for research into how we might diagnose and treat diseases using these potent genetic and microbial insights.
“For hundreds of thousands of years, humans have co-evolved with the bacteria that live within and on us. This has resulted in a finely tuned equilibrium between our bacteria, genes, food, and health that is still difficult to untangle and research to this day.
Investigating very big datasets of several thousand people to robustly uncover common relationships is one viable technique to shed additional insight on this remarkable association. Several other recent research involving people from different parts of the world have already confirmed our findings.
This collection of studies, taken together, exemplifies the complex link that exists between humans and their gut microbial ecosystems, as well as highlighting crucial health consequences “Dr. Meric came to a conclusion.


canadian pharmaceuticals https://canadadrugs.pro/# – prescription drug prices canadadrugs.pro
legitimate canadian mail order pharmacies https://canadadrugs.pro/# – online pharmacy no scripts canadadrugs.pro
canadian pharmacy meds reviews https://canadianinternationalpharmacy.pro/ ordering drugs from canada
canada pharmacy online https://canadianinternationalpharmacy.pro/ canada cloud pharmacy
best ed pills online https://edpill.cheap/ treatments for ed
cipro ciprofloxacin https://cipro.guru/ ciprofloxacin 500mg buy online
antibiotics cipro https://cipro.guru/ antibiotics cipro
cipro for sale https://cipro.guru/ ciprofloxacin 500mg buy online
can i buy diflucan over the counter https://diflucan.pro/ diflucan 6
https://doxycycline.auction/
lana rhoades modeli – https://lanarhoades.fun/ lana rhoades
sweety fox https://sweetiefox.online/ – Sweetie Fox video
Sweetie Fox filmleri https://sweetiefox.online/ – Sweetie Fox video
Sweetie Fox modeli https://sweetiefox.online/ – Sweetie Fox filmleri
Sweetie Fox filmleri https://sweetiefox.online/ – Sweetie Fox video
swetie fox https://sweetiefox.online/ – sweeti fox
sweeti fox https://sweetiefox.online/ – Sweetie Fox izle
Sweetie Fox filmleri https://sweetiefox.online/ – Sweetie Fox modeli
2KdCX8 sweetie fox video https://sweetiefox.pro/ – sweetie fox full video
L5p9C8 sweetie fox full video https://sweetiefox.pro/ – ph sweetie fox
fox sweetie https://sweetiefox.pro/ – sweetie fox full video
eva elfie new video https://evaelfie.site/ eva elfie new video
tRL5bm fox sweetie https://sweetiefox.pro/ – sweetie fox full video
Bfh7T7 sweetie fox new https://sweetiefox.pro/ – sweetie fox cosplay
eva elfie full video https://evaelfie.site/ eva elfie full videos
eva elfie hot https://evaelfie.site/ eva elfie full video
mia malkova photos: https://miamalkova.life/ mia malkova hd
mia malkova only fans: https://miamalkova.life/ mia malkova only fans
mia malkova girl: https://miamalkova.life/ mia malkova hd
mia malkova full video: https://miamalkova.life/ mia malkova girl
mia malkova only fans: https://miamalkova.life/ mia malkova photos
mia malkova full video: https://miamalkova.life/ mia malkova full video
mia malkova hd: https://miamalkova.life/ mia malkova full video
aviator hilesi: https://aviatoroyunu.pro aviator sinyal hilesi
aviator bet https://aviatorjogar.online/ – jogar aviator online
aviator bet https://aviatorjogar.online/ – pin up aviator
aviator bet https://aviatormalawi.online – aviator
aviator betting game https://aviatormalawi.online – aviator bet malawi login
aviator https://aviatormalawi.online – aviator game online
aviator betting game https://aviatormalawi.online – aviator bet
aviator bet malawi https://aviatormalawi.online – aviator game online
aviator bet https://aviatormalawi.online – aviator game online
aviator bet malawi https://aviatormalawi.online – aviator
aviator malawi https://aviatormalawi.online – aviator bet malawi
play aviator: https://aviatorghana.pro/ play aviator
aviator game: https://aviatorghana.pro/ aviator game online
aviator ghana: https://aviatorghana.pro/ aviator game
aviator login: https://aviatorghana.pro/ play aviator
pin-up casino login https://pinupcassino.pro/ cassino pin up
aviator login: https://aviatorghana.pro/ aviator betting game
aviator game online: https://aviatorghana.pro/ aviator betting game
cassino pin up https://pinupcassino.pro/ pin-up cassino
pin-up https://pinupcassino.pro/ pin up aviator
pin up https://pinupcassino.pro/ cassino pin up
aviator oficial pin up https://pinupcassino.pro/ pin-up casino
site de apostas – https://jogodeaposta.fun/ melhor jogo de aposta para ganhar dinheiro
aviator jogo de aposta – https://jogodeaposta.fun/ jogo de aposta
jogo de aposta online – https://jogodeaposta.fun/ aviator jogo de aposta
aviator online https://aviatormocambique.site aviator moçambique
aviator online https://aviatormocambique.site jogar aviator
aviator bahis: https://aviatoroyunu.pro aviator
gates of olympus oyna ücretsiz – https://gatesofolympus.auction/ gate of olympus hile
gates of olympus demo – https://gatesofolympus.auction/ gates of olympus demo
gates of olympus – https://gatesofolympus.auction/ gates of olympus 1000 demo
gates of olympus demo – https://gatesofolympus.auction/ gates of olympus taktik
gate of olympus hile – https://gatesofolympus.auction/ gates of olympus nasıl para kazanilir
gates of olympus sırları – https://gatesofolympus.auction/ gates of olympus oyna
gates of olympus giriş – https://gatesofolympus.auction/ gates of olympus demo türkçe
güncel sweet bonanza https://sweetbonanza.bid/ – sweet bonanza taktik
sweet bonanza indir https://sweetbonanza.bid/ – sweet bonanza yasal site
sweet bonanza nasıl oynanır https://sweetbonanza.bid/ – sweet bonanza siteleri
bonus veren casino slot siteleri https://slotsiteleri.guru/ deneme bonusu veren slot siteleri
slot siteleri https://slotsiteleri.guru/ slot oyun siteleri
slot casino siteleri https://slotsiteleri.guru/ güvenilir slot siteleri
deneme veren slot siteleri https://slotsiteleri.guru/ slot oyunları siteleri
en iyi slot siteleri https://slotsiteleri.guru/ slot oyun siteleri
en çok kazandiran slot siteleri https://slotsiteleri.guru/ casino slot siteleri
pin-up giriş: https://pinupgiris.fun/ pin up casino güncel giriş
pin up casino güncel giriş: https://pinupgiris.fun/ pin-up giriş
pin up aviator: https://pinupgiris.fun/ pin up bet
pin-up casino indir: https://pinupgiris.fun/ pin-up bonanza
aviator oyunu 20 tl https://aviatoroyna.bid – aviator oyna 100 tl
aviator hilesi https://aviatoroyna.bid – aviator uçak oyunu
aviator oyunu 20 tl https://aviatoroyna.bid – aviator hile
aviator sinyal hilesi apk https://aviatoroyna.bid – aviator bahis
aviator hilesi ücretsiz https://aviatoroyna.bid – aviator hilesi ücretsiz
aviator https://aviatoroyna.bid – aviator uçak oyunu
uçak oyunu bahis aviator https://aviatoroyna.bid – aviator oyna 100 tl
aviator https://aviatoroyna.bid – aviator hile
best india pharmacy: reputable indian online pharmacy buy medicines online in india indianpharmacy.icu
top 10 pharmacies in india: canadian pharmacy india indian pharmacy paypal indianpharmacy.icu
top 10 pharmacies in india: reputable indian online pharmacy india online pharmacy indianpharmacy.icu
reputable indian online pharmacy: indian pharmacies safe online pharmacy india indianpharmacy.icu
mail order pharmacy india: indianpharmacy com top online pharmacy india indianpharmacy.icu
indian pharmacy: buy medicines online in india best online pharmacy india indianpharmacy.icu
mexican pharmaceuticals online https://mexicanpharmacy.shop/# mexican border pharmacies shipping to usa mexicanpharmacy.shop
mexican rx online http://mexicanpharmacy.shop/# mexico drug stores pharmacies mexicanpharmacy.shop
mexican rx online https://mexicanpharmacy.shop/# buying prescription drugs in mexico mexicanpharmacy.shop
mexican rx online https://mexicanpharmacy.shop/# buying from online mexican pharmacy mexicanpharmacy.shop
canadian pharmacy india: canadian pharmacy 24h com safe canadian neighbor pharmacy canadianpharmacy24.store
legitimate canadian pharmacies: canadian pharmacy service canada pharmacy reviews canadianpharmacy24.store
canadian neighbor pharmacy: canadian pharmacy near me canadian pharmacy king canadianpharmacy24.store
canadian discount pharmacy: northwest canadian pharmacy best canadian pharmacy canadianpharmacy24.store
reddit canadian pharmacy: canada cloud pharmacy best canadian pharmacy canadianpharmacy24.store
canadian pharmacy online reviews: medication canadian pharmacy canada pharmacy online canadianpharmacy24.store
canadian valley pharmacy: canadianpharmacymeds canadian pharmacies that deliver to the us canadianpharmacy24.store
canadian mail order pharmacy https://canadianpharmacy24.store/# – canadian pharmacy 1 internet online drugstore canadianpharmacy24.store
casino trực tuyến việt nam https://casinvietnam.shop/# – chơi casino trực tuyến trên điện thoại
casino trực tuyến uy tín https://casinvietnam.com/# – đánh bài trực tuyến
casino trực tuyến việt nam http://casinvietnam.shop/# – đánh bài trực tuyến
cost of propecia without prescription https://finasteride.store/ order propecia without insurance
buying propecia without rx https://finasteride.store/ buy propecia for sale
buying generic propecia for sale https://finasteride.store/ cost generic propecia for sale
cipro 500mg best prices https://ciprofloxacin.tech/ where can i buy cipro online
buy ciprofloxacin over the counter https://ciprofloxacin.tech/ cipro
ciprofloxacin 500 mg tablet price https://ciprofloxacin.tech/ cipro pharmacy
where can i buy cipro online https://ciprofloxacin.tech/ cipro
ciprofloxacin mail online https://ciprofloxacin.tech/ cipro generic